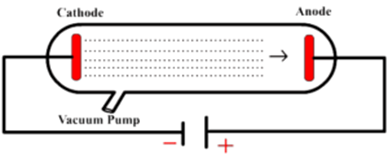
His first experiment was to build a cathode ray tube with a metal cylinder on the end. Thomson had an inkling that the ‘rays’ emitted from the electron gun were inseparable from the latent charge, and decided to try and prove this by using a magnetic field. 
Thomson constructed some elegant experiments to find a definitive and comprehensive answer about the nature of cathode rays. They debated whether the rays were waves or particles, as they seemed to exhibit some of the properties of both. Physicists knew that the ray carried a negative charge but were not sure whether the charge could be separated from the ray.  
However, other researchers, notably Crookes, argued that the focused nature of the beam meant that they had to be particles. With more experimentation, researchers found that the ‘cathode rays’ emitted from the cathode could not move around solid objects and so traveled in straight lines, a property of waves. William Crookes discovered that a tube coated in a fluorescing material at the positive end, would produce a focused ‘dot’ when rays from the electron gun hit it. Later and improved cathode ray experiments found that certain types of glass produced a fluorescent glow at the positive end of the tube. This cathode ray also became known as an ‘electron gun’. Physicists in the 19th century found out that if they constructed a glass tube with wires inserted in both ends, and pumped out as much of the air as they could, an electric charge passed across the tube from the wires would create a fluorescent glow. Look at any glowing neon sign or any ‘old-fashioned’ television set, and you are looking at the modern descendants of the cathode ray tube. What is a Cathode Ray Tube?Įven without consciously realizing it, most of us are already aware of what a cathode ray tube is. This research led to further experiments by Bohr and Rutherford, leading to an understanding of the structure of the atom. His better-known research proved the existence of negatively charged particles, later called electrons, and earned him a deserved Nobel Prize for physics. Scholarly yet accessible to those with basic physics knowledge, this book should be of interest to historians of science, professional scientists and engineers, teachers and students of physics, and general readers interested in the development of modern physics.Like most scientists of that era, he inspired generations of later physicists, from Einstein to Hawking. The author also focuses on the controversies over N-rays, Becquerel's positive electron, and the famous Ehrenhaft-Millikan dispute over subelectrons. It explores the events leading to Millikan's unambiguous isolation of the electron and the simultaneous circumstances surrounding the birth of Ernest Rutherford's nuclear atom and the discovery of radioactivity in 1896. It also provides an overview of the history of modern physics up to the advent of the old quantum theory around 1920.Ĭonsolidating scholarly material while incorporating new material discovered by the well-respected author, the book covers the continental and English race for the source of the cathode rays, culminating in Thomson's corpuscle in 1897. The book traces the evolution of the concept of electrical charge, from the earliest glow discharge studies to the final cathode ray and oil drop experiments of J.J. Thomson's Electron presents the compelling story of the discovery of the electron and its role as the first subatomic particle in nature. Flash of the Cathode Rays: A History of J.J. The electron is fundamental to almost all aspects of modern life, controlling the behavior of atoms and how they bind together to form gases, liquids, and solids.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
